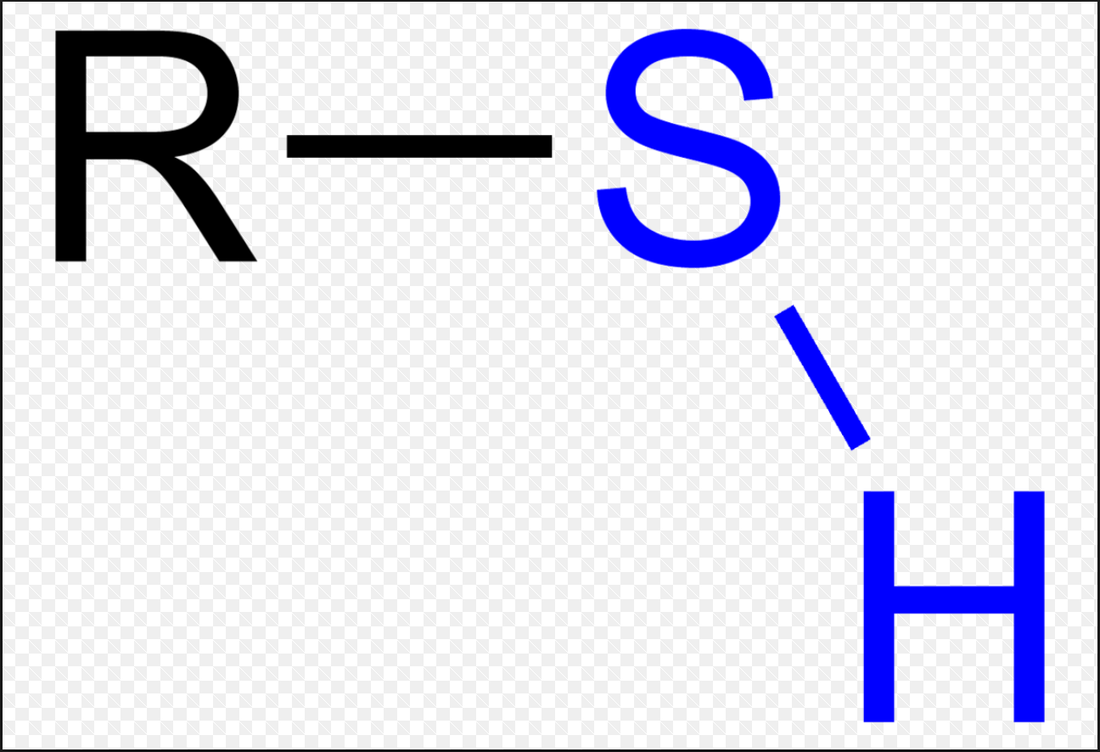ORGANIC COMPOUNDS
Functional Groups
|
There are 7 functional groups:
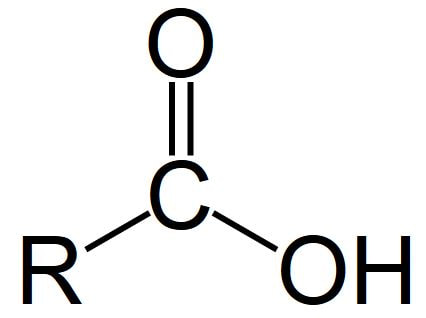
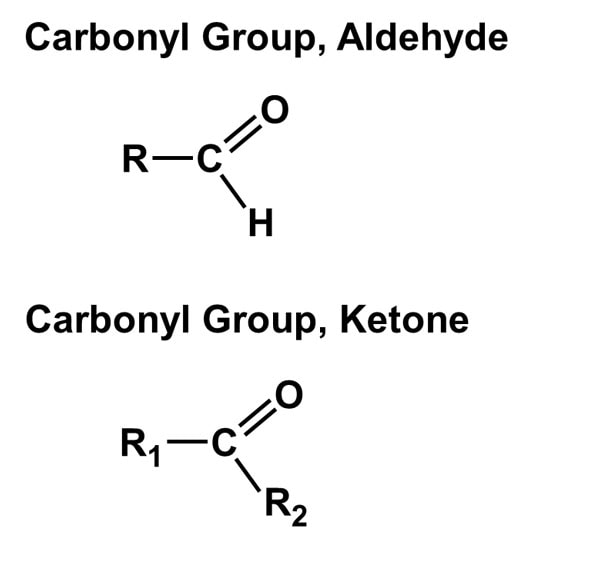
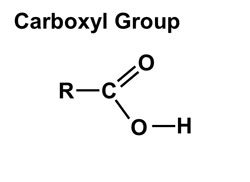
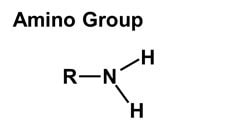
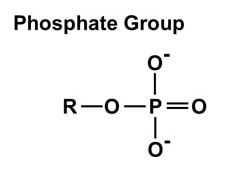
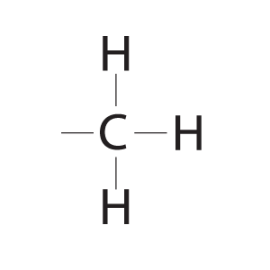
1. Hydroxyl group (-OH): Molecules with hydroxyl groups are generally referred to as alcohols, and are easily soluble in water. Examples include sugars and alcohols. 2. Carbonyl group (-C=O): If an organic molecule has this group at the end of the molecule, it is referred to as an aldehyde (like glucose). If it's in the middle, it is referred to as a ketone (like fructose). 3. Carboxyl group (-COOH): This group, present in all amino acids and proteins, contributes to the acidity of the molecule. Examples include acetic acid and formic acid. 4. Amino group (-NH2): This group, present in all amino acids, proteins, and some specific nucleotides, can act as a base by accepting protons. This group contributes to the basic nature of molecules. 5. Sulfhydryl group (-SH): This group is often found within proteins to stabilize their structures. They are also present in some active sites involved in the catalysis of enzymes. 6. Phosphate group (-OPO3): This group is important in energy compounds, such as ATP and ADP. They are also present in DNA and RNA, and phospholipids. This group is highly reactive and contributes to the acidity of a molecule. 7. Methyl group (-CH3): This group is non-polar and is significant in determining the solubility of a compound in aqueous solutions. Methylation involves the addition of a methyl group, and can make a molecule non-functional. This group is found in alcohols and fatty acids. |